All the emulsions had monomodal oil droplet size distribution and wider ranges were directly related to bigger sizes, especially at low (10 mg/mL) protein concentration when compared to the 50 mg/mL.Īlbumin Flaxseed Globulin Protein solubility SDS-PAGE Surface hydrophobicity.Ĭopyright © 2018 Elsevier Ltd. which amino acids with nonpolar, hydrophobic R groups cluster together on the inside of the protein, leaving hydrophilic amino acids on the outside to interact with surrounding water molecules. Amino acid analysis showed significantly (p < 0.05) higher levels of hydrophobic amino acids in the globulin, which was consistent with higher surface hydrophobicity when compared to the albumin. 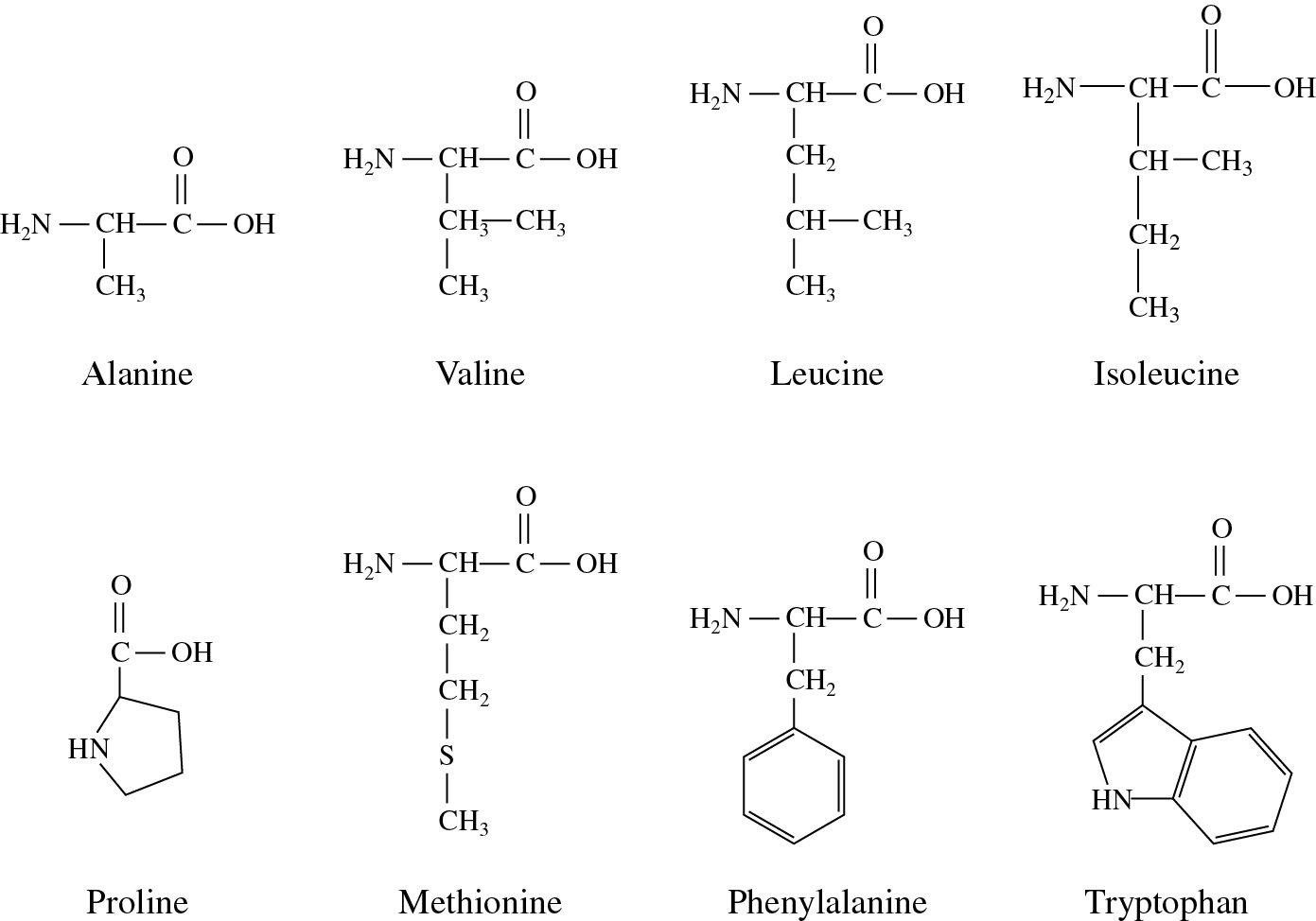 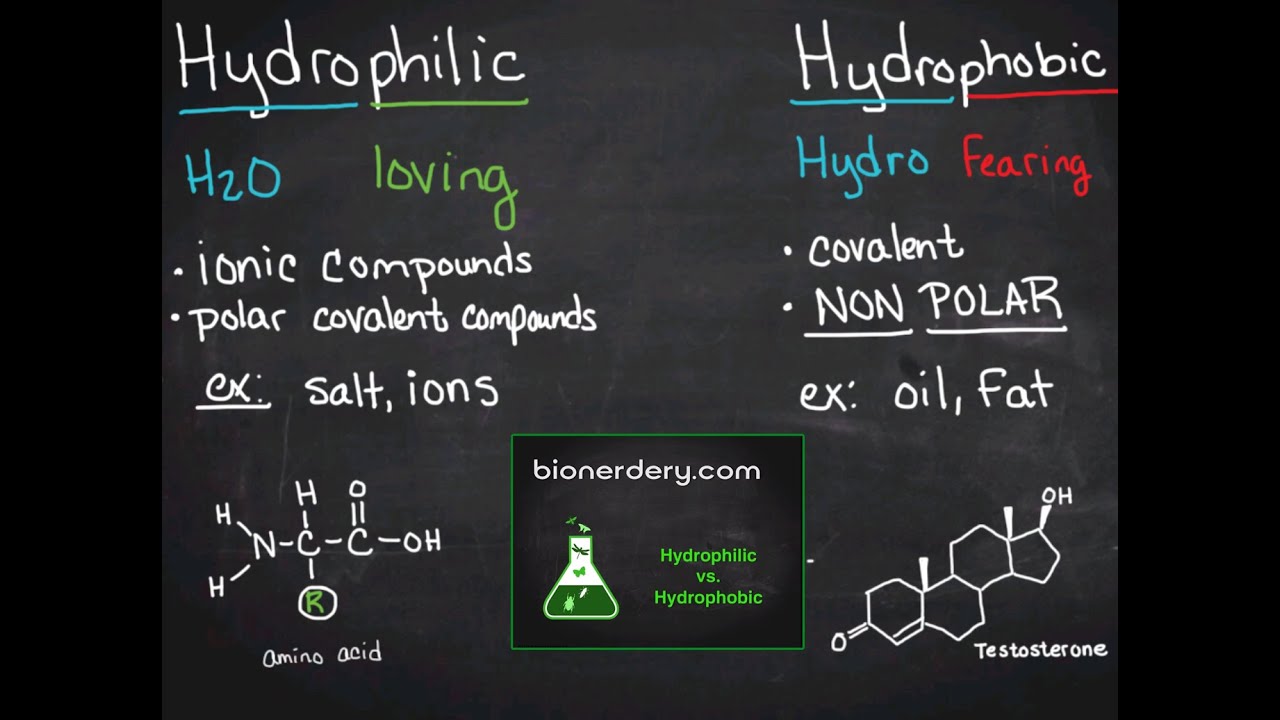
Gel electrophoresis data indicate that the globulin is composed of several polypeptides in the 10-50 kDa range while albumin consisted mainly of the 10 kDa polypeptide accompanied by a minor content of 40 kDa. Amino acid sequence analysis of the purified proteins confirmed the successful conversion from procolipase to colipase. The opposite of a nonpolar molecule is, as you might guess, polar. You know how oil and water don't mix That's because oil is hydrophobic. Flaxseed protein meal was extracted with 0.5 M NaCl, and the extract dialyzed against water followed by centrifugation to obtain the globulin as a water-insoluble precipitate and albumin as the water-soluble albumin. The nonpolar molecules we'll be talking about are hydrophobic amino acids, meaning 'water fearing' because they don't mix with water molecules. The physicochemical and emulsification characteristics of flaxseed albumin and globulin protein fractions were determined in this study. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
January 2023
Categories |
 RSS Feed
RSS Feed
